Polycyclic Aromatic Hydrocarbons (PAHs)
Polycyclic aromatic hydrocarbons (PAHs) are a class of organic compounds that consist solely of carbon and hydrogen atoms in aromatic ring structures. Sixteen PAHs are regulated by the U.S. Environmental Protection Agency (USEPA) based on their potential human and ecological health effects. These compounds can be naturally occurring (e.g., forest fires) or anthropogenic (e.g., coal gasification, automobile exhaust). Remedial techniques are available for addressing PAH-contaminated soil, groundwater, and surface waters. However, such efforts must carefully consider the hydrophobic nature of PAHs, effects of PAH weathering in soil/sediment, and the poor biodegradability of high-molecular weight PAHs. Bioavailability of PAHs is also a key consideration for health and ecological risk assessment and selection of remedial techniques. Here, we review the physical and chemical properties of PAHs, their toxicity and rationale as priority pollutants, remedial options, and risk assessment considerations.
Related Article(s):
- Biodegradation - Hydrocarbons
- Monitored Natural Attenuation (MNA) of Fuels
- Petroleum Hydrocarbons (PHCs)
- Remediation Technologies
Contributor(s): Dr. Stephen Richardson
Key Resource(s):
- Polycyclic aromatic hydrocarbons chemistry and carcinogenicity[1]
- Provisional guidance for quantitative risk assessment of polycyclic aromatic hydrocarbons[2]
Introduction
Polycyclic aromatic hydrocarbons (PAHs), also known as polyaromatic hydrocarbons, are a class of hundreds of organic compounds that consist of two or more aromatic rings fused in linear, angular, or clustered arrangements[1]. PAHs are ubiquitous in the environment, predominantly formed by the incomplete combustion of organic materials from both natural sources (e.g., forest fires, volcanic events), and anthropogenic activities (e.g., coal gasification, automobile exhaust, incinerators, coke production, cooking, tobacco smoke)[1]. The USEPA has listed 16 PAHs as 'priority pollutants' in aquatic and terrestrial ecosystems[3]. Seven of these PAHs may cause cancer in humans[2], and benzo[a]pyrene is considered the highest cancer risk amongst the 16 PAHs[2][4].
Physical and Chemical Properties
PAHs are hydrophobic and do not readily dissolve in water or volatilize to the atmosphere (with the exception of naphthalene, which was once used in 'moth balls'). The chemical stability, low water solubility, and high sorption capacity of PAHs contribute greatly to their persistence in the environment[5][6]. PAHs can be divided into two categories: (1) low molecular weight PAHs composed of less than four aromatic rings (e.g., naphthalene, acenaphthene, fluorene, phenanthrene), and (2) high molecular weight PAHs composed of four or more rings (e.g., pyrene, chrysene, benzo[a]pyrene, dibenz[a,h]anthracene). High molecular weight PAHs are generally less water soluble, have lower vapor pressures and Henry’s constants, and partition more readily into organic matter than low molecular weight PAHs. Selected physical and chemical properties of the 16 USEPA-regulated PAHs are presented in Table 1.
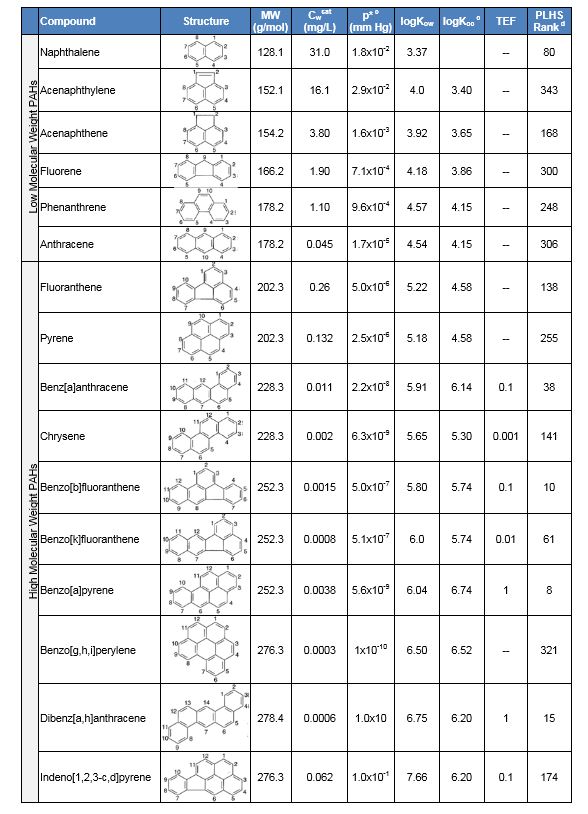
Toxicity
The most important property driving PAH remediation is their toxicity (or carcinogenicity). PAHs ranked 9th on the 2015 Agency for Toxic Substances and Disease Registry (ATSDR) Priority List of Hazardous Substances (PLHS) based on their toxicity, frequency of occurrence at USEPA National Priorities List (i.e. Superfund) sites, and potential for human exposure[10]. Individually, all 16 regulated PAHs are included on the ATSDR PLHS, with six ranked in the top 100; most notably, benzo[a]pyrene at 8th (Table 1).
Regulatory guidelines or site-specific cleanup goals for soil commonly account for PAH toxicity by assigning toxic equivalency factors (TEFs) to individual PAHs (Table 1), normalized to benzo[a]pyrene toxicity. Multiplying the measured concentration of each PAH by its respective TEF yields an equivalent concentration of benzo[a]pyrene for the PAH mixture, called a benzo[a]pyrene equivalent[4]. These adjusted values often serve as soil remediation goals at PAH-contaminated sites. For drinking water, the USEPA has established a maximum contaminant level for benzo[a]pyrene of 0.2 µg/L.
PAH Bioavailability
Bioavailability is an important concept for PAH remediation and risk assessment in soil and sediments. With respect to bioremediation, bioavailability refers to the contaminant fraction that can be effectively accessed by contaminant-degrading microbial communities[8][11]. Bioavailability and degradation of PAHs in natural soils and sediment are generally two-phased, with an initial phase of rapid PAH removal followed by a longer period of limited PAH reduction. During the initial phase, bioavailability of PAHs is high and degradation rates may be limited by reaction rate (e.g., microbial uptake rather than mass transfer from soil particles). As bioavailable PAHs are removed, mass transfer mechanisms (desorption and diffusion) become controlling factors for the rate of PAH degradation[12]. Physical, chemical, and biological remedial methods have been developed to either increase PAH bioavailability (e.g., mixing, surfactants, cosolvents), or decrease PAH bioavailability (e.g., biostabilization, sediment capping, solidification), depending on the treatment design and site cleanup goals[13]. The former generally incorporate a mass removal step (e.g., enhanced biodegradation, aqueous phase extraction) to minimize risk. The latter strategies reduce risk by creating a barrier between the contaminations and surrounding receptors.
Remediation of PAHs
Anthropogenic sources of PAHs vary widely, including former manufactured gas plants, petroleum fuel spills, coal- and gas-fired power plants, and industrial incinerators, as well as are present in a range of environments (e.g., harbor sediments, marine waters), often proximate to past or current industrial operations. The fate and transport of PAHs in these environments is controlled by PAH hydrophobicity, rates of dissolution, physicochemical properties of the soil, and the source phase. In sediments, for example, PAHs sorb to natural organic matter and are present in oils, tars, residues, and other nonaqueous phase liquids (NAPLs) deposited from industrial activities. These phases act as long-term sources of PAHs to the water column through dissolution processes and can greatly influence overall PAH transport, degradation, and bioavailability[14]. A variety of ex situ and in situ remediation methods have been used to address PAH contaminated soils, groundwater, and surface waters. The most common ex situ practices include landfill disposal, incineration, thermal desorption, and soil washing[15]. Generally, these methods are expensive and can be cost-prohibitive for sites with large footprints, significant depth of contamination, and existing infrastructure. Alternatively, in situ treatments such as chemical oxidation, solvent and surfactant flushing, and bioremediation are available, although they are used to a lesser extent than ex situ methods. These remediation methods are generally less expensive, but require longer treatment times to meet regulatory criteria.
Physical treatments such as in situ stabilization and solidification prevent or minimize the release (or leaching) of PAHs from contaminated soils and sediments by using binding agents (e.g., cement, asphalt, fly ash, and clay) to limit water infiltration and bind PAHs into less mobile forms. The addition of granular activated carbon to contaminated sediments has also been tested as a means to strongly bind available PAHs and reduce the ecological risks to overlying surface waters[16][17].
Chemical treatments for PAHs include Fenton’s reagent, hydrogen peroxide, activated persulfate, and ozone[18]. These methods generate very reactive free radicals (e.g., hydroxyl radical, sulfate radical, ozone radical) and other reactive species (e.g., persulfate anion, peroxides), capable of attacking the aromatic structure of PAHs. Advantages of in situ chemical oxidation over conventional remediation methods include reasonable treatment times, reactivity with a broad range of PAHs, and destruction of contaminants in situ. However, the use of chemical oxidants is complicated by oxidation of non-target species such as soil organic matter and limited control of oxidant delivery in heterogeneous media[18]. Biodegradation of PAHs can occur both aerobically and anaerobically in the subsurface. Numerous aerobic bacterial species and fungi are capable of transforming two-, three-, and four-ring PAHs to non-toxic end products such as water and carbon dioxide (i.e., PAH mineralization) and partially degrading five- and six-ring PAHs to intermediate compounds[19]. Under anaerobic conditions, degradation of two- and three-ring PAHs has been documented under nitrate[20][21], iron[22], and sulfate-reducing conditions[23][24]. However, rates of anaerobic PAH biodegradation are generally much lower (several orders of magnitude) than aerobic metabolism. Since many contaminated sites are oxygen- and nutrient-limited, a variety of biostimulation methods (e.g., can be composting, landfarming, biosparging, peroxide injection) can be used to deliver oxygen/nutrients into groundwater and soil/sediments to stimulate aerobic degradation of PAHs[25][26][27][28].
References
- ^ 1.0 1.1 1.2 Harvey, R.G., 1991. Polycyclic aromatic hydrocarbons: chemistry and carcinogenicity. Cambridge University Press: Cambridge, 396 pgs. ISBN 978-0521292047
- ^ 2.0 2.1 2.2 U.S. Environmental Protection Agency, 1993. Provisional guidance for quantitative risk assessment of polycyclic aromatic hydrocarbons. EPA 600-R-93-089. Report pdf
- ^ Keith, L. and Telliard, W., 1979. ES&T special report: priority pollutants: I-a perspective view. Environmental Science & Technology, 13(4), 416-423. doi: 10.1021/es60152a601
- ^ 4.0 4.1 LaGoy, P.K., Quirk, T.C., 1994. Establishing generic remediation goals for the polycyclic aromatic hydrocarbons: critical issues. Environmental Health Perspectives, 102(4), 348-352. Report pdf
- ^ Kanaly, R.A., Harayama, S., 2000. Biodegradation of high-molecular-weight polycyclic aromatic hydrocarbons by bacteria. Journal of Bacteriology, 182(8), 2059-2067. doi: 10.1128/JB.182.8.2059-2067.2000
- ^ Van Hamme, J.D., Singh, A., Ward, O.P., 2003. Recent advances in petroleum microbiology. Microbiology and Molecular Biology Reviews, 67(4), 503-549. doi: 10.1128/MMBR.67.4.503-549.2003
- ^ LaGrega, M.D., Buckingham, P.L., Evans, J.C., 2001. Hazardous waste management: 2nd edition. McGraw-Hill, Boston. ISBN 1577666933.
- ^ 8.0 8.1 National Research Council (US). Committee on Bioavailability of Contaminants in Soils and Sediments, 2003. Bioavailability of contaminants in soils and sediments: Processes, tools, and applications. Washington, DC: National Academies Press., 432 pgs. doi: 10.17226/10523
- ^ Mackay, D., Shiu, W.Y., Ma, K.C., 1997. Illustrated handbook of physical-chemical properties of environmental fate for organic chemicals.
- ^ ATSDR, 2015. Comprehensive environmental response, compensation, and liability act (CERCLA) priority list of hazardous substances. List
- ^ Ortega-Calvo, J.J., Harmsen, J., Parsons, J.R., Semple, K.T., Aitken, M.D., Ajao, C., Eadsforth, C., Galay-Burgos, M., Naidu, R., Oliver, R., Peijnenburg, W.J., Römbke, J., Streck, G., Versonnen, B. 2015. From bioavailability science to regulation of organic chemicals. Environmental Science & Technology, 49(17), 10255-10264. doi: 10.1021/acs.est.5b02412
- ^ Bosma, T.N., Middeldorp, P.J., Schraa, G., Zehnder, A.J., 1997. Mass transfer limitation of biotransformation: quantifying bioavailability. Environmental Science & Technology, 31(1), 248-252. doi: 10.1021/es960383u
- ^ Ehlers, L.J., Luthy, R.G., 2003. Contaminant bioavailability in soil and sediment. Environmental Science & Technology, 37, 295A-302A. doi: 10.1021/es032524f
- ^ Wick, A.F., Haus, N.W., Sukkariyah, B.F., Haering, K.C., Daniels, W.L., 2011. Remediation of PAH-contaminated soils and sediments: a literature review. Virginia Polytechnic Institute and State University, USA. Report pdf
- ^ U.S. Environmental Protection Agency, 2004. Cleaning up the nation's waste sites: markets and technology trends. EPA 542-R-04-015. Report pdf
- ^ Luthy, R.G., Zimmerman, J.R., McLeod, P.B., Zare, R.N., Mahajan, T., Ghosh, U., Bridges, T.S., Millward, R.N., Talley, J.W., 2004. In situ stabilization of persistent organic contaminants in Marine Sediments. Strategic Environmental Research and Development Program, Arlington, Virginia. SERDP Project ER-1207. ER-1207
- ^ Luthy, R.G., Zimmerman, J.R., McLeod, P.B., Zare, R.N., Mahajan, T., Ghosh, U., Bridges, T.S., Millward, R.N., Talley, J.W., 2014. Demonstration of in situ treatment with reactive amendments for contaminated sediments in active DoD harbors. Project ER-201131. Strategic Environmental Research and Development Program, Arlington, Virginia. ER-201131
- ^ 18.0 18.1 Huling, S. G., Pivetz, B. E., 2006. In-situ chemical oxidation (No. EPA/600/R-06/072). Environmental Protection Agency Washington DC Office of Water. Report pdf
- ^ Cerniglia, C.E., 1992. Biodegradation of polycyclic aromatic hydrocarbons. Biodegradation, 3(2-3), 351-368. doi: 10.1007/BF00129093
- ^ Mihelcic, J.R., Luthy, R.G., 1988. Degradation of polycyclic aromatic hydrocarbon compounds under various redox conditions in soil-water systems. Applied and Environmental Microbiology, 54(5), 1182-1187. Journal Article Page
- ^ McNally, D.L., Mihelcic, J.R., Lueking, D.R., 1998. Biodegradation of three-and four-ring polycyclic aromatic hydrocarbons under aerobic and denitrifying conditions. Environmental Science & Technology, 32(17), 2633-2639. doi: 10.1021/es980006c
- ^ Anderson, R.T., Lovley, D.R., 1999. Naphthalene and benzene degradation under Fe(III)-reducing conditions in petroleum-contaminated aquifers. Bioremediation Journal, 3(2), 121-135. doi: 10.1080/10889869991219271
- ^ Coates, J.D., Anderson, R.T., Woodward, J.C., Phillips, E.J., Lovley, D.R., 1996. Anaerobic hydrocarbon degradation in petroleum-contaminated harbor sediments under sulfate-reducing and artificially imposed iron-reducing conditions. Environmental Science & Technology, 30(9), 2784-2789. doi: 10.1021/es9600441
- ^ Coates, J.D., Anderson, R.T., Lovley, D.R., 1996. Oxidation of polycyclic aromatic hydrocarbons under sulfate-reducing conditions. Applied and Environmental Microbiology, 62(3), 1099-1101. Journal Article
- ^ Mueller, J.G., Chapman, P.J., Pritchard, P.H., 1989. Creosote-contaminated sites. Their potential for bioremediation. Environmental Science & Technology, 23(10), 1197-1201. doi: 10.1021/es00068a003
- ^ Johnson, C.R., Scow, K.M., 1999. Effect of nitrogen and phosphorus addition on phenanthrene biodegradation in four soils. Biodegradation, 10(1), 43-50. doi: 10.1023/A:1008359606545
- ^ Breedveld, G.D., Sparrevik, M., 2000. Nutrient-limited biodegradation of PAH in various soil strata at a creosote contaminated site. Biodegradation, 11(6), pp.391-399. doi: 10.1023/A:1011695023196
- ^ Carmichael, L.M., Pfaender, F.K., 1997. The effect of inorganic and organic supplements on the microbial degradation of phenanthrene and pyrene in soils. Biodegradation, 8(1), 1-13. doi: 10.1023/A:1008258720649